-
COVID-19 Convalescent Plasma (CCP)
Stanford Blood Center (SBC) has received FDA approval under a Biologics License Application (BLA) to manufacture and distribute high-titer COVID-19 Convalescent Plasma (CCP) nationwide. SBC is the first blood center in California and only the second in the United States to achieve this milestone. This full licensure replaces the previous Emergency Use Authorization (EUA) and [...] More -
Frozen Plasma
Plasma separated from whole blood by centrifugation (200 mL product, anticoagulant CPD) or collected by a centrifugation apheresis technique (400 mL product, anticoagulant ACD-A). Frozen at -18C within 8 hours of collection (6 hours for ACD-A plasma), FFP contains plasma proteins including all coagulation factors. Volume ~ 200 or 400 mL Fibrinogen 2-4 mg/mL Coagulation …
More -
Recovered Plasma, Frozen
Plasma separated from whole blood by centrifugation and frozen at -18C between 8 and 24 hours of whole blood collection. Contains Factor VIII:C as well as other labile and stable coagulation factors, anticoagulant CPD. Volume ~200 mL Factor VIII:C ~150 IU Storage, frozen < -18C, 12 months Storage, thawed 1-6C, 24 hours
More -
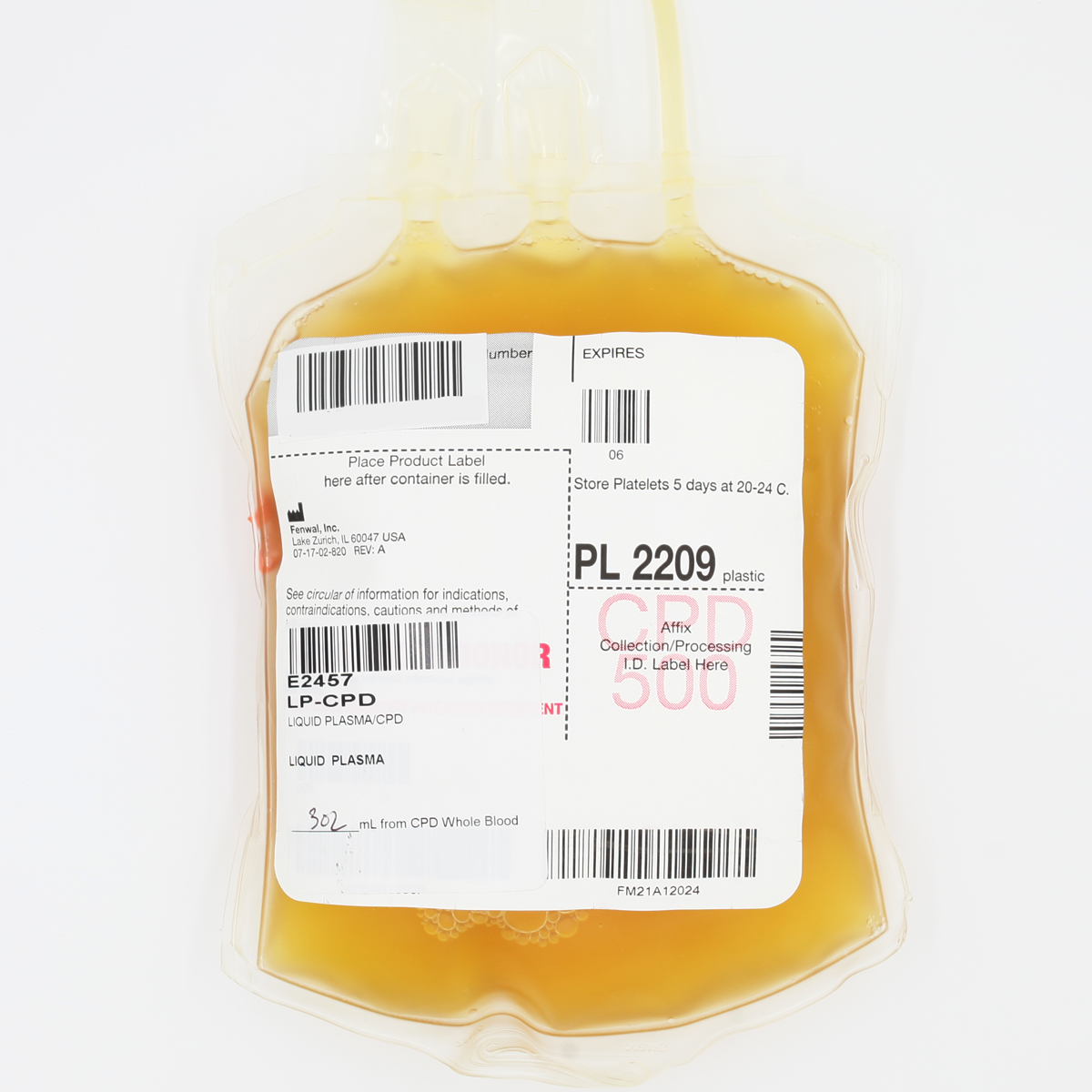
Plasma, Liquid
Fluid portion of whole blood separated by centrifugation from whole blood unit (200 mL product, anticoagulant CPD) or by apheresis (400 mL product, anticoagulant ACD-A). Contains plasma proteins including stable coagulation factors. Volume 200 or 400 mL Fibrinogen ~2-4 mg/mL Stable coagulation factors only* 1 IU/mL Storage 1-6C, 24 hours
More